Establishment of an irritation assessment system specifically for the development of formulations for delicate areas
: Introduction of a safety assessment system using a three-dimensional cultured human vaginal mucosal epithelium model.
December 18, 2025
ROHTO Pharmaceutical Co., Ltd. (Headquarters: Osaka City, President: Hidetoshi Segi) is working on product development and safety design utilizing alternative methods to animal testing in order to realize ROHTO Group Comprehensive Management Vision 2030, "Connect for Well-being." This time, with the aim of evaluating the safety of a cleansing agent specifically for delicate areas, the company constructed an irritation evaluation system using a three-dimensional cultured human vaginal mucosal epithelium model and confirmed the safety of its self-developed product in both in vitro and human actual use tests. The results of this research were presented at the 38th Annual Meeting of the Japanese Society for Alternatives to Animal Experiments, held from November 1st to 3rd, 2025.
Key points of the research
- In the unexplored area of "delicate parts" where established in vitro evaluation methods do not exist, we designed a unique evaluation process and applied it to actual product development.
- Using a three-dimensional cultured human vaginal mucosal epithelium model, we predict the irritation level of cleansers specifically designed for delicate areas.
- We hope that research will be promoted with a view to establishing safety evaluation standards for Feminine Care products, including the creation of guidelines and external collaborations.
Research Background
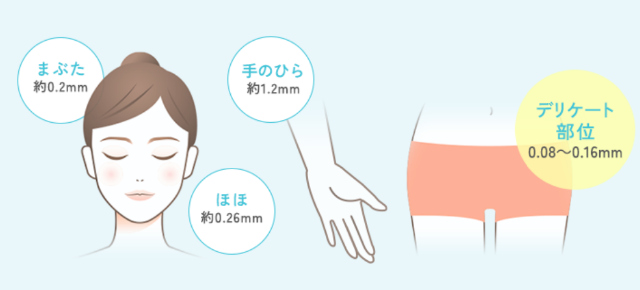
Figure 1: Skin thickness in different parts of the body
In recent years, interest in Feminine Care products has been growing to address concerns such as odor, dryness, and itching in delicate areas. However, because the skin in delicate areas is very thin (Figure 1), has a weak barrier function, and is adjacent to mucous membranes, careful consideration of the risk of irritation is necessary.
Therefore, the development of Feminine Care products requires more careful safety design than general skin products, but there are no standardized in vitro testing methods specifically for delicate areas, and establishing an evaluation system that can be used in product development has been a challenge.
Therefore, in this study, we designed a new process for evaluating the irritancy of delicate areas by using a "three-dimensional cultured human vaginal mucosal epithelial model" reconstructed from human vulva-derived cells and independently investigating exposure conditions and evaluation methods, and incorporated this into product development.
result
Result 1: Irritation evaluation using a three-dimensional cultured human vaginal mucosal epithelial model.
In this study, we used our company's cleansing agents specifically for delicate areas and for sensitive skin as test samples. All of these formulations were confirmed to be free from skin irritation issues in prior safety tests (Table 1).
When the irritancy of a cleanser specifically for delicate areas was evaluated using a three-dimensional cultured human vaginal mucosal epithelium model, the cell viability was high at 94.8±0.7%, suggesting low irritancy. In addition, the cell viability of a cleanser for sensitive skin under the same test conditions was 80.9±2.4%, indicating a tendency for the cleanser specifically for delicate areas to have a higher cell viability (Figure 2). Furthermore, when the irritancy of the cleanser specifically for delicate areas and the cleanser for sensitive skin was evaluated using a three-dimensional reconstructed human epidermis model, which is commonly used for evaluating irritancy in healthy skin, the cell viability was high at 107.2±1.7% and 102.4±8.4%, respectively, confirming low irritancy. No significant difference in irritancy was observed between the cleanser specifically for delicate areas and the cleanser for sensitive skin.

Table 1: Summary of test samples (our company's cleaning agent) and various safety test results.
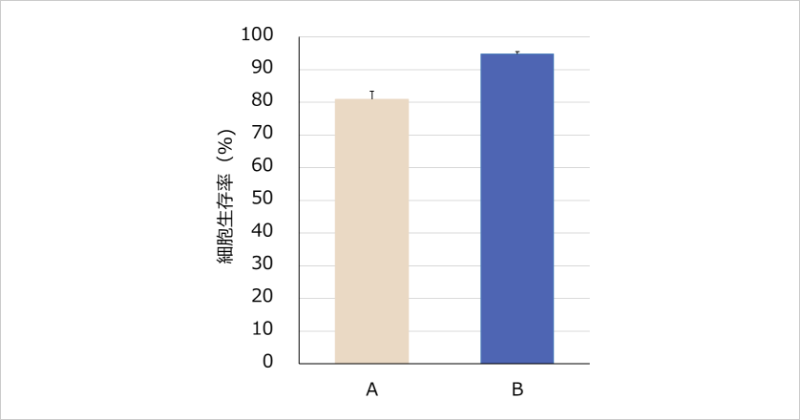
Figure 2: Comparative evaluation of cell viability in a three-dimensional cultured human vaginal mucosal epithelial model (n=3)
<Testing Method>
The test substance was added to a three-dimensional cultured human vaginal mucosal epithelium model and incubated for a predetermined time.
Subsequently, the model was washed with PBS, and cell viability (%) was calculated based on the absorbance (OD value) obtained by the MTT method. Cell viability was determined by dividing the average OD value of the test substance by the average OD value of the negative control (distilled water), and multiplying the ratio by 100 (n=3).
(Conducted by Rohto Pharmaceutical Research Institute)
Result 2: Safety confirmed in human use trials
A real-world usage test was conducted on 25 women aged 21 to 49, in which they used a cleanser specifically for delicate areas for four weeks. The results confirmed that it was "almost safe to safe."
<How to use>
The subjects used a cleanser specifically for delicate areas at least once a day during bathing for four weeks. They took an appropriate amount (1-2 pumps), gently washed the delicate area *, and rinsed thoroughly with water or lukewarm water.
* Delicate areas: Female genitalia and surrounding areas (including the outside of the vagina)
<Evaluation Method>
Observations by a dermatologist were conducted at the start of continuous use of the test product and after 4 weeks of continuous use. Based on the occurrence of side effects, a comprehensive safety evaluation of the delicate area cleanser was performed for each subject's test site.
Consideration
The results of this study suggest that the three-dimensional cultured human vaginal mucosal epithelium model is more sensitive to irritation than the three-dimensional reconstructed human epidermis model. Furthermore, when compared with the safety evaluation results from human clinical trials, the in vitro evaluation using the three-dimensional cultured human vaginal mucosal epithelium model shows potential as a pre-evaluation method prior to human safety confirmation in the development of formulations specifically for delicate areas.
Future outlook
Based on the results of this research, we aim to establish a more practical and reliable in vitro evaluation system and widely apply it to product development that prioritizes quality and safety, with the customer at the center. Furthermore, we intend to take the lead in safety evaluation of Feminine Care products used on delicate areas, with a view to collaborating with external organizations and developing guidelines.