Successful construction of a next-generation vascular-attached artificial skin model with cellular diversity
– inventing a new anti-aging theory that focuses on how cellular diversity generates skin elasticity.
April 14, 2026
ROHTO Pharmaceutical Co., Ltd. (Headquarters: Osaka City, President: Hidetoshi Segi), in collaboration with Professor Tatsuya Shimizu of the Institute for Advanced Biomedical Engineering and Science, Tokyo Women's Medical University, has constructed a next-generation vascular-attached artificial skin that autonomously reproduces the complex "cellular diversity" of living skin.
In this model, we successfully cultured fibroblasts and perivascular cell-like cell populations specific to living skin, which were difficult to maintain using conventional culture methods, and achieved skin barrier function and elasticity close to that of living skin. Furthermore, we revealed part of the mechanism by which vitamin C improves signs of skin aging through perivascular cells. These results are expected to accelerate the development of next-generation skincare products and pharmaceuticals as an ethical and highly accurate evaluation system to replace animal testing.
The results of this research were published in the online scientific journal "EMBO Reports" (April 1st).
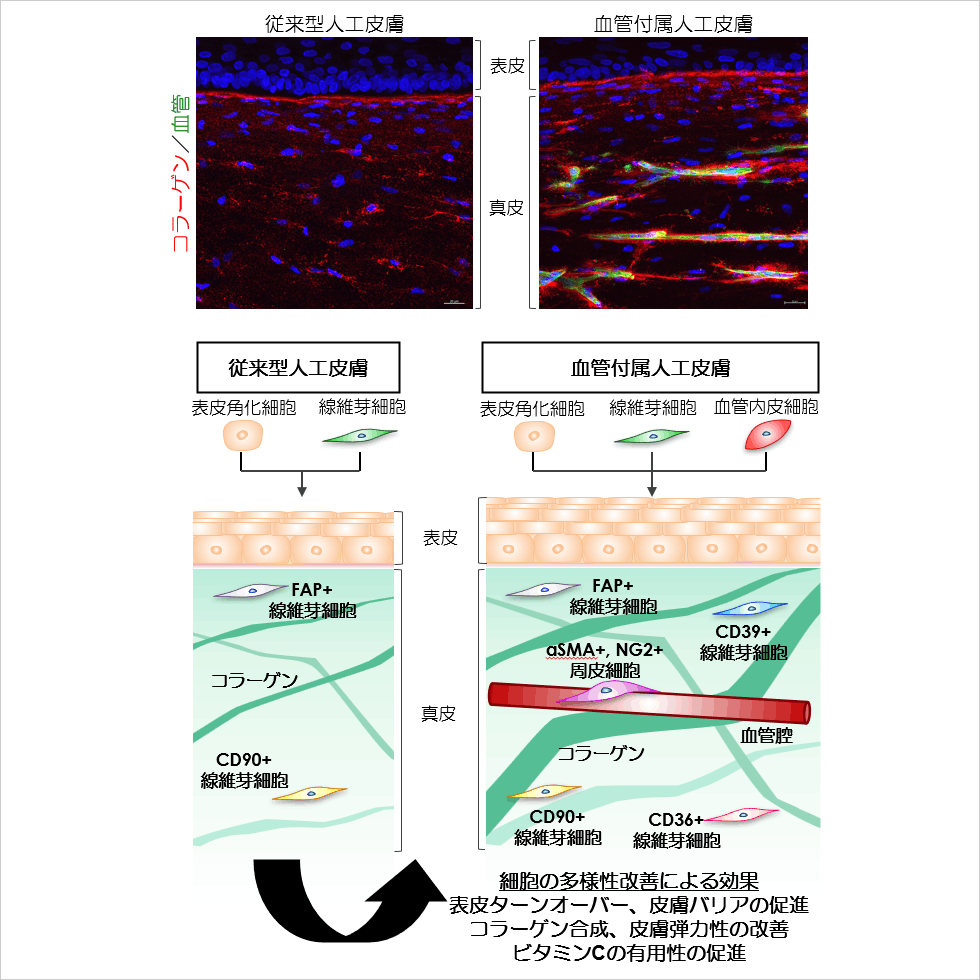
Figure 1. By adding blood vessels, an artificial skin was developed that highly replicates the diversity and function of human dermal mesenchymal cells.
Key points of the research
- We developed artificial skin that replicates the diversity of dermal cells similar to those in living organisms through co-culturing with vascular endothelial cells.
- We discovered that increased cellular diversity improves the skin's barrier function and elasticity.
- We discovered that in skin with cellular diversity, the effects of vitamin C on improving skin barrier function and skin elasticity are enhanced.
Research Background
The cells that make up the skin have been broadly classified according to their shape and function, such as epidermal keratinocytes and dermal fibroblasts. In recent years, with the development of techniques such as single-cell gene expression analysis, it has become clear that even within the same type of cell, there are differences in function and properties that can be described as individuality. Interestingly, it has become clear that the individuality of dermal mesenchymal cells is lost with age, and this is beginning to be considered an important factor in skin aging and disease. However, conventional cell culture techniques have not been able to reproduce or analyze this cellular diversity, making it difficult to prove the relationship with aging and to construct care theories. Furthermore, in response to the global trend of reducing animal testing, there is a need for advanced alternative testing methods that can mimic the complexity of living organisms.
Therefore, in this study, we hypothesized that blood vessels play a crucial role in maintaining cellular diversity, and began constructing an artificial skin model that replicates human cellular diversity by building artificial skin with attached blood vessels. Furthermore, by comparing the presence or absence of blood vessels in the artificial skin, we investigated how vascularized cellular diversity affects the skin.
result
Construction of artificial skin that replicates the diversity of dermal mesenchymal cells by introducing vascular endothelial cells.
Conventional artificial skin is a two-layer model of the epidermis and dermis, consisting of two types of cells: epidermal keratinocytes and dermal fibroblasts. First, the research team constructed an artificial skin model with a vascular structure using three types of cells: epidermal keratinocytes, dermal fibroblasts, and umbilical cord-derived vascular endothelial cells (Figure 1). Next, single-cell RNA sequencing analysis was performed on the cells contained in the constructed artificial skin to evaluate the properties and diversity of the cells contained in the dermis. The dermis of living human skin contains diverse fibroblasts and vascular pericytes, but the diversity was lost in cells cultured in a planar manner for research purposes. On the other hand, it was confirmed that the diversity distribution pattern of fibroblasts and vascular pericytes in the constructed artificial skin was very similar to that of living human skin (Figure 2). This result indicates that the three-dimensional environment and intercellular interactions between blood vessels and skin play an important role in reproducing the cellular heterogeneity of fibroblasts and vascular pericytes, which has been difficult to reproduce until now.

Figure 2. Comparison of dermal mesenchymal cell diversity in human skin and artificial skin using single-cell RNA sequencing.
[Testing Method]
Using three types of cells—normal human epidermal keratinocytes, normal human dermal fibroblasts, and human umbilical cord vascular endothelial cells—we constructed three samples: ① cultured cells of the three types on a petri dish, ② a conventional artificial skin constructed from two types of cells (epidermal keratinocytes and dermal fibroblasts), and ③ a vascular-attached artificial skin incorporating vascular endothelial cells into the conventional artificial skin. Single-cell RNA sequencing analysis was performed on the cells of each sample. The obtained comprehensive gene expression profile data was integrated with publicly available human skin cell data (Ref. Solé-Boldo et al. Single-cell transcriptomes of the human skin reveal age-related loss of fibroblast priming. Commun Biol 3, 188 (2020)), and principal component analysis was performed. Furthermore, cells with similar gene expression were classified, and the results were plotted in two dimensions on UMAP to visualize cell populations with similar gene expression (conducted by Rohto Pharmaceutical Research Institute).
Improvement of skin barrier function and dermal elasticity through the introduction of vascular endothelial cells.
Next, we compared the functionality of artificial skin with and without blood vessels. As a result, improvements were observed in skin's moisturizing barrier function, turnover function (which plays an important role in transparency), and skin elasticity (related to wrinkles and sagging) with the addition of blood vessels (Figure 3).
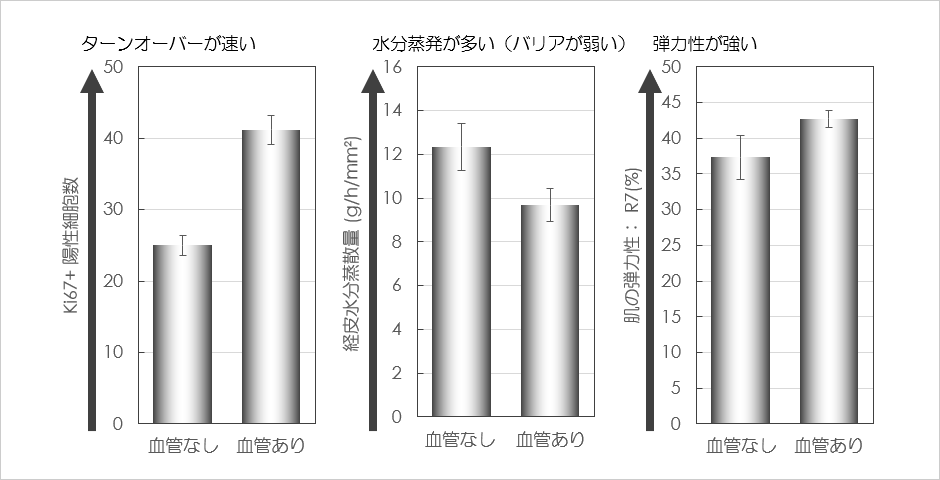
Figure 3. Functional changes in an artificial skin model due to vascularization.
[Testing Method]
Conventional artificial skin without blood vessels and artificial skin with blood vessels were constructed, and their epidermal turnover, barrier function, and dermal elasticity were analyzed. Epidermal turnover was measured by counting the number of Ki67-positive epidermal basal cells using immunohistochemical analysis. Transepidermal water loss was measured using Tewameter™ HEX. Skin elasticity was measured using the R7 value with Cutometer MPA 580 (conducted by Rohto Pharmaceutical Research Institute).
The effects of vitamin C in improving cell turnover and skin elasticity are enhanced by artificial skin attached to blood vessels.
Furthermore, we compared the responsiveness of artificial skin to vitamin C with and without blood vessels. As a result, we confirmed that the usefulness of vitamin C was enhanced by the addition of blood vessels in relation to the skin's moisturizing barrier function, turnover function which plays an important role in transparency, and skin elasticity related to wrinkles and sagging (Figure 4).
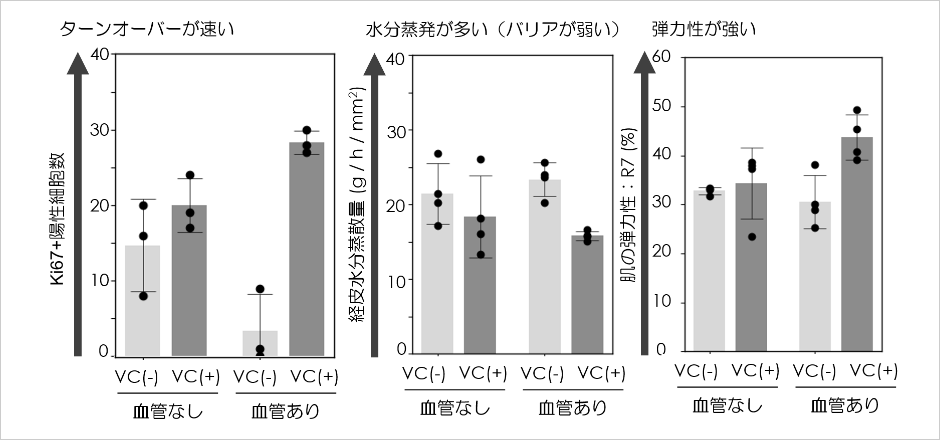
Figure 4. Changes in the response to vitamin C in an artificial skin model induced by vascularization.
[Testing Method]
Conventional artificial skin without blood vessels and artificial skin with blood vessels were constructed and cultured for a certain period using a culture medium containing or without vitamin C. Epidermal turnover, barrier function, and dermal elasticity were then analyzed. Epidermal turnover was measured by counting the number of Ki67-positive epidermal basal cells using immunohistochemical analysis. Transepidermal water loss was measured using Tewameter™ HEX. Skin elasticity was measured using the R7 value with a Cutometer MPA 580 (conducted by Rohto Pharmaceutical Research Institute).
Future outlook
This study revealed that the presence of vascular endothelial cells induces fibroblast diversity, playing a crucial role in maintaining skin structure and function, as well as in drug responsiveness. This research suggests the existence of a mechanism by which skin aging and disease are controlled by vascular development and regression, supporting a new skincare theory that focuses on blood vessels.
Furthermore, because the developed artificial skin model exhibits physiological responses closer to those of human skin than conventional models, we believe it can become a powerful alternative to animal testing in the search for highly functional skincare ingredients that improve wrinkles and sagging, as well as in the evaluation of the safety and efficacy of pharmaceuticals.
Paper Information
<Title>
The heterogeneity of dermal mesenchymal cells reproduced in skin equivalents regulates barrier function and elasticity
<Author Name>
Shun Kimura, Sachiko Sekiya, Sawa Yamashiro, Tetsutaro Kikuchi, Masatoshi Haga, & Tatsuya Shimizu
<Magazine>
EMBO Reports
<DOI>
https://doi.org/10.1038/s44319-026-00757-w
Terminology
*1: Single-cell RNA sequencing
Next-generation sequencing is a method that comprehensively examines the entire mRNA, or the overall picture of gene expression, contained within individual cells. Conventional RNA sequencing extracts mRNA from all cells in a sample and examines gene expression levels, so it only reveals the average gene expression of the cell population. Single-cell sequencing makes it possible to examine the differences in gene expression of individual cells, which can be considered the individuality of each cell. Using this technique, it has been discovered that certain cell populations within seemingly identical cells play a role in controlling various life phenomena.
*2: Immunohistochemical analysis method
This method uses antibodies capable of binding to specific proteins to stain and visualize target proteins in tissues. In this study, we are using antibodies against proteins such as type 1 collagen, a major component of the dermis; CD31, which is characteristically present in vascular endothelial cells; and Ki67, which is present in epidermal keratinocytes during cell proliferation associated with cell turnover, to investigate changes in tissue condition and skin function.